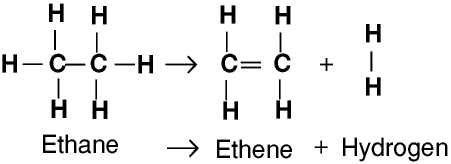
This is only one way in which this particular molecule might break up. Or, showing more clearly what happens to the various atoms and bonds: One possible reaction involving the hydrocarbon C 15H 32 might be: The hydrocarbon molecules are broken up in a fairly random way to produce mixtures of smaller hydrocarbons, some of which have carbon-carbon double bonds. There isn't any single unique reaction happening in the cracker. These fractions are obtained from the distillation process as liquids, but are re-vaporised before cracking. The source of the large hydrocarbon molecules is often the naphtha fraction or the gas oil fraction from the fractional distillation of crude oil (petroleum). This is achieved by using high pressures and temperatures without a catalyst, or lower temperatures and pressures in the presence of a catalyst. 
This page describes what cracking is, and the differences between catalytic cracking, thermal cracking and steam cracking used in the petrochemical industry.Ĭracking is the name given to breaking up large hydrocarbon molecules into smaller and more useful bits.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
